What Is the Risk of Having an Abnormal Baby From a Robertsonian Carrier
- Case Report
- Open Admission
- Published:
A family with Robertsonian translocation: a potential mechanism of speciation in humans
Molecular Cytogenetics volume 9, Article number:48 (2016) Cite this commodity
Abstract
Groundwork
Robertsonian translocations occur in approximately one in every 1000 newborns. Although nearly Robertsonian translocation carriers are good for you and accept a normal lifespan, they are at increased risk of spontaneous abortions and risk of producing unbalanced gametes and, therefore unbalanced offspring. Here nosotros reported a previously undescribed Robertsonian translocation.
Case Presentation
We identified 3 Robertsonian translocation carriers in this family unit. Two were heterozygous translocation carriers of 45,Twenty or XY,der(14;fifteen)(q10;q10) and their son was a homozygous translocation carrier of a 44,XY,der(14;15)(q10;q10), der(14;15)(q10;q10) karyotype. Chromosomal analysis of sperm showed 99.7 % of sperm from the homozygous translocation carrier were normal/balanced while merely 79.9 % of sperm from the heterozygous translocation carrier were normal/counterbalanced. There was a significantly higher frequency of aneuploidy for sex chromosome in the heterozygous translocation carrier.
Conclusions
The reproductive fitness of Robertsonian translocation carriers is reduced. Robertsonian translocation homozygosity can be a potential speciation in humans with 44 chromosomes.
Background
Robertsonian translocations, fusions between two acrocentric chromosomes, are the well-nigh common structural chromosomal rearrangements in humans and occur in approximately 1 in every chiliad newborns [1]. It occurs in the five acrocentric chromosomes, 13, 14, 15, 21, and 22, which have very pocket-sized short artillery that contain no unique genes. During a Robertsonian translocation, the participating chromosomes break at their centromeres and the long arms fuse to grade a single chromosome with a single centromere. The brusk arms also join to class a smaller reciprocal production, which typically contains nonessential genes and is ordinarily lost inside a few cell divisions. This blazon of translocation is cytologically visible and can reduce chromosome number if the smaller chromosome that results from a translocation is lost over cellular divisions. Notwithstanding, the smaller chromosome lost may carry and so few genes that it can be lost without an ill effect to the individual [two, 3].
When the translocation is balanced, the person with it is chosen a Robertsonian translocation carrier. As carriers are healthy and have a normal lifespanand, many never discover their unusual chromosome rearrangement. Therefore, the translocation tin be passed downwardly in families for many generations without anyone discovering. Robertsonian translocation carriers produce six types of gametes. At the end of meiosis I, segregation of the translocated and nontranslocated chromosomes from the two different chromosome pairs involved leads to the formation of either counterbalanced gametes via alternate segregation or unbalanced gametes via adjacent segregation during anaphase [1, two]. Meiotic tetravalent configuration tends to segregate in alternating way, resulting in preferential production of normal/counterbalanced spermatozoa. However, certain percentages of unbalanced gametes derived from next segregation are likewise produced, leading to the increased run a risk of miscarriage and chromosomally unbalanced fetus. In this report, we investigated a Robertsonian translocation family and analyzed the meiotic segregation patterns and interchromosomal furnishings in sperm from two Robertsonian translocation carriers.
Case presentation
Clinical information
A 25-year-onetime Chinese man (4-i) married to a not-consanguineous woman with normal chromosomes (4-2). This couple had a son (V-one) who died at 6-month old without an autopsy. Previous chromosome assay indicated V-one had a 45,XY,der(fourteen;fifteen)(q10;q10) karyotype. The parents of the propositus (IV-1) are phenotypically normal first cousins (Iii-1, Iii -ii). Their parents, a common uncle and both grandparents were non available for cytogenetic analysis (Fig. 1). Mental state test was performed on IV-i past a psychiatrist.
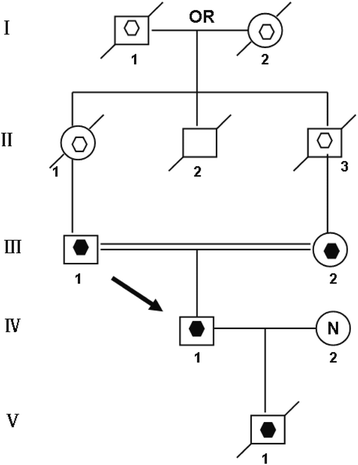
Pedigree of the family transmitting Rob translocation chromosome t(fourteen; 15)(q10;q10) Open hexagon designates a presumed carrier of t(14; 15)(q10;q10). Filled hexagon designates a known carrier of t(14;15)(q10;q10). The proband, IV-1 (pointer), has disomy t(fourteen;15)(q10;q10). The proband's married woman, Four-two, had a normal karyotype. Their deceased son, V-one, was a carrier of karyotype 45,XY,der(14;15)(q10;q10)
Karyotype assay
G-banding karyotype analysis was performed on all the iv members in the family. The previously undescribed karyotype, 44,XY,der(14;15)(q10;q10), der(14;15)(q10;q10), was further authenticated by the Chinese Academic Committee of the State Key Laboratory of Medical Genetics (Fig. ii).
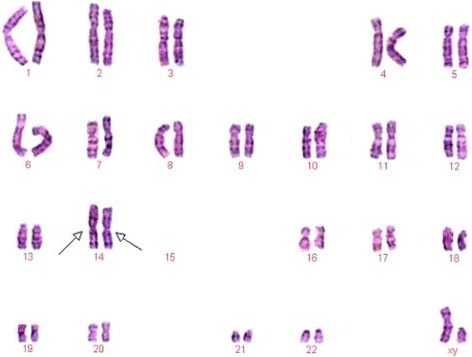
Karyotype of the proband with 44,XY,der(14;fifteen)(q10;q10),der(fourteen;15)(q10;q10) chromosomal constitution The arrows indicated disomy of chromosome der(fourteen;xv)
Fluorescence in situ hybridization (FISH)
Sperm chromosomes from patient 3-one and 4-i were analyzed by FISH. Semen samples were assessed for volume, concentration and motion according to the Globe Health Organization criteria. After semen analysis, progressively mobile sperm were isolated and washed twice in phosphate buffered saline (pH vii.4) and centrifuged at 1500 rpm for eleven min. Pellets were fixed with 5 ml of acetic acid/methanol mixture (i:iii) for at least 30 min at iv °C. Aliquots (twoscore–l μl) of the resulting suspension of nuclei were smeared on cold pre-cleaned slides. Nuclei decondensation was performed in 1 N NaOH for 2 min. The slides were then degraded past a graded ethanol series (lxx %, ninety %, 100 %) and denatured with 0.25 % formamide in 2xSSC followed by overnight hybridization with a combination of commercially available probes. Two sets of probe mixtures were used in this study. Dual-colour FISH was carried out using locusspecific probes (LSP) and Tel probes from Vysis (Vysis, Downers Grove, IL, USA). For patient IV-1 and 3-1,TelVysion Probe 14q (D14S1420, Spectrum Red) was used for 14q32.33 and TelVysion Probe 15q(D15S120, Spectrum Green) was used for 15q26.three.
To investigate the presence of interchromosomal outcome, triple-color FISH was performed using the second probe mixture, which consists of commercial satellite (DNA) probes from Vysis, including chromosomes 18, X and Y (CEP 18, Spectrum Blue/CEP 10, Spectrum Light-green/CEP Y, Spectrum Scarlet). Postal service-hybridization washes included ii min in 0.4xSSC/0.3%NP-40 (pH = seven) at 72 °C, followed by ane min in 2xSSC/0.1%NP-40 (pH = 7) at room temperature. Slides were covered with DAPI II (Vysis). Only intact spermatozoa bearing a like degree of decondensation and articulate hybridization signals were scored. All disrupted or overlapping spermatozoa were excluded from analysis. Slides with hybridization efficiency of 99 % and more were analyzed. 1,000 sperm nuclei were analyzed per patient.
Assortment comparative genomic hybridization (aCGH)
Assortment comparative genomic hybridization (aCGH) has been introduced in clinical diagnosis to rapidly detect genomewide gains and losses with higher resolution [four]. It is a loftier throughput method which detects re-create number changes to a resolution of even as low as 1 Kb. For aCGH analysis, DNA was extracted from peripheral claret [v]. aCGH assay was performed on using the viii × threescore K commercial arrays (Agilent Technologies, Santa Clara, CA, United states).
Data assay
The statistical analysis was performed using the SAS system (2002–2003, SAS Plant Inc., Cary, NC). Chi-squared test was used for meiotic segregation assay. A probability value of less than 0.05 (P < 0.05) was considered to exist statistically significant.
Results
The karyotype analysis was performed on four members in the family and detected two dissimilar karyotypes of Robertsonian translocation. The results are shown in below. III-i and Iii-2 were heterozygous while Four-one was homozygous.
-
III-1: 45,XY,der(xiv;15)(q10;q10)
-
III-two: 45,XX,der(14;15)(q10;q10)
-
Four-ane: 44,XY,der(14;15)(q10;q10), der(xiv;xv)(q10;q10)
-
IV-two: 46,XX
Because the patient (IV-1) has a symptom of apathy, we referred him to see a psychiatrist for a thorough mental status examination. The patient had no psychiatric history and remained cooperative during the psychiatric interview. His thought processes were linear, logical, and consistently goal directed and his judgment was intact. Therefore, no psychiatric symptoms were observed. In improver, neurology consultation revealed no focal neurological deficits. The results of Craniocerebral Computed Tomography (CT) and lumbar puncture (LP) were normal. Electroencephalography (EEG) showed no signs of seizure activity.
The aCGH assay on Iv-one detected a 2.03 Mb duplication in the region of q11.i-q11.two on chromosome 15 (Fig. 3). Co-ordinate to the Copy number variation database (CNVdb), this duplication is a chromosomal polymorphism in normal population. Because IV-one is a is Homologous Robertsonian translocation carrier and his two derivative chromosomes came from the aforementioned Robertsonian translocation carrier, we speculated that each of the two derivative chromosomes should accept a ii.03 Mb duplication in the region of q11.1-q11.two on chromosome xv.

aCGH analysis of IV-ane revealed a 2.03 Mb duplication at chromosome 15 q11.1-q11.2 (arr 15q11.1q11.2(xx,481,702-22,509,254)x4)
To assay the sperm of 3-1 and IV-1, semen samples from the two different Robertsonian translocation carriers (Three-i and Iv-ane) were assessed for book, concentration and move according to the World Health Organization criteria. The volume, concentration and mobility of sperm in 3-one are significantly reduced due to his sometime age. In contrast, they are all normal in Iv-1 (Table 1). We farther analyzed the meiotic segregation patterns in these two dissimilar Robertsonian translocation carriers. In the heterozygote Robertsonian translocation carrier (Three-1), the frequency of normal/counterbalanced spermatozoa resulting from alternating segregation was 79.nine % while the frequency of unbalanced spermatozoa resulting from adjacent segregation was 20.1 %, which is significantly college than normal population [five–8] (Table two). In contrast, in the homozygote Robertsonian translocation (IV-ane), the frequency of balanced spermatozoa was 99.vii % while the frequency of unbalanced spermatozoa was but 0.iii %, which is similar to normal populations [5–8] (Table 2). These information demonstrated there nearly all sperm of Robertsonian translocation homozygotes were balanced while those of heterozygotes had a per centum of unbalanced. In addition, chromosome assay of 4-1'southward sperm showed either a 22,X,der (xiv;15) karyotype or 22,Y,der(14;15) karyotype supporting our speculation. As shown in Fig. 4, the arrow pointed a sperm with four fluorescence signals (2 red and two dark-green) suggesting this sperm has two derivative chromosomes of der(14;15). These data suggest a potential mechanism of speciation in humans via Robertsonian translocation.
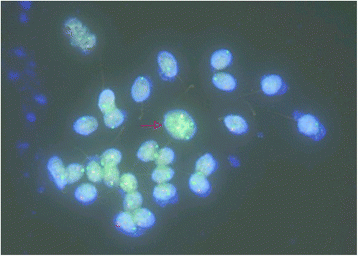
Sperm cells after hybridization with TelVysion 14q32.33 (Spectrum Red) and TelVysion 15q26.3 (Spectrum Green). The sperm with crimson arrow may be a diploid sperm
The influence of translocated chromosomes on the synapses and disjunction of other chromosomes is called an interchromosomal effect (Water ice) [9]. To determine whether ICE was present, the nullisomy, disomy and diploid rates for chromosomes 18, X and Y were analyzed in both Robertsonian translocation carriers (Table 3). In the heterozygote Robertsonian translocation carrier (III-ane), in that location was a pregnant increase in the rate of sex chromosome aneuploidy and diploid. In contrast, in the homozygote Robertsonian translocation (IV-1), the incidence of spermatozoa with nullisomy, disomy and diploid for the sexual activity chromosomes remained similar to normal populations (Table 3) [v–8]. This information suggest that Water ice only presents in certain Robertsonian translocation carriers.
Give-and-take
Rob translocation carries are phenotypically normal since they involve loss only of brusque arm fabric. Many such rearrangments in natural populations of unlike species are known. Robertsonian rearrangments can increase polymorphisms in a species, provide material for natural selection and even lead to speciation [10, 11]. It has been argued that the types of chromosome rearrangments that occur as polymorphisms or as fixed permanent heterozygotes invariably involve meta- or submetacentric chromosomes. Those that distinguish species and serve to isolate these species involve telocentric or acrocentric chromosomes, which are self-sterilizing [12]. In our written report, Iv-1, who is a homologous Robertsonian translocation carrier, was healthy and had a counterbalanced chromosomal complement. Assessment of a semen sample from showed normal sperm morphology and motility. More chiefly, 99.7 % of his sperm are normal or balanced (Table 2) and there was no interchromosomal effect (Tabular array 3).
Multicolor FISH on decondensed sperm nuclei allows for a rapid analysis of meiotic segregation in sperm of translocation carriers, providing information on the exact corporeality of normal/balanced sperm [xiii]. Such information is undoubtedly important not only for basic cytogenetic inquiry just likewise for reproductive counseling of Robertsonian translocation carriers. t(14;15) are rare Robertsonian translocations. Our data confirmed previous studies showing that alternating segregation was more often than next segregations in Robertsonian translocations carriers [5, 8]. However, the meiotic segregation patterns and interchromosomal effects in ii Robertsonian translocation carriers are unlike (Table ii). These findings are consequent with previously published data [14–16]. The loftier prevalence of the alternate segregation is presumably due to cis-configuration of the trivalent during meiosis, which favors an alternate segregation in all Robertsonian translocations [ii]. Although a Robertsonian translocation carrier has a full genetic complement, their productive fettle is reduced due to high probability of genetically imbalanced gametes.
The interchromosomal result could be explained past the formation of heterosynapses between chromosomes involved in the translocation and the sex vesicle, which could also involve other chromosomes [17]. Ice remains controversial in the literature. Some publications have indicated its presence with Robertsonian translocation [xviii, 19] while others demonstrated no evidence of this phenomenon [4, 20]. In our report, nosotros observed higher incidence of aneuploidy for sex chromosomes in spermatozoa in the heterozygote Robertsonian translocation just not the homozygote Robertsonian translocation suggesting that ICE only presents in some Robertsonian translocation carriers.
Robertsonian rearrangements are common chromosomal changes that can lead to rapid and efficient reproductive isolation between karyotypically similar populations, especially when many Robertsonian metacentric chromosomes display monobrachial homologies [10]. Homozygous Robertsonian translocations take reported by several groups [21–23] and systemically reviewed by the O'Neill group [24]. Dallapiccola et al. [21] reported a fetus with two t(14;21) chromosomes. The related parents were heterozygous for the same translocation. Martinez et al. [22] reported three adult siblings homozygous for t(13;xiv). Their parents were beginning-cousins and both were heterozygous carriers. Rajangam et al. [23] found a unique DS karyotype 45, XY, der(xiv;21)pat, der(14;21)mat, +21mat. Although translocation heterozygosity is associated with meiotic disturbances, which volition lead to infertility and subfecundity, it should non have any effect on meiosis. Here nosotros reported a homo with a 44,XY,der(14;xv)(q10;q10),der(14;15)(q10;q10) karyotype. Robertsonian translocation may provide material for evolution. Long term isolation of a group of individuals who are homozygous for a particular Robertsonian translocation chromosome could theoretically lead to the establishment of a new man subspecies having a full genetic complement in 44 chromosomes. Although karyotypic differences between species have long been recognized, the question of whether these mutations play a causal role in speciation remains unanswered [25–27]. In our written report, all sperm of Robertsonian translocation homozygotes were counterbalanced while those of heterozygotes had a percentage of unbalanced ones implicating of a potential of the homozygosity of Robertsonian translocations for speciation.
Conclusions
To conclude, in this study we reported a previously described Robertsonian translocation karyotype. The homozygosity of Robertsonian translocation for speciation may be a potential mechanism of speciation in Humans.
Abbreviations
aCGH, Array comparative genomic hybridization; CT, Craniocerebral Computed Tomography; EEG, Electroencephalography; FISH, Fluorescence in situ hybridization;; Water ice, interchromosomal effect; LP, lumbar puncture
References
-
Therman E, Susman B, Denniston C. The nonrandom participation of human acrocentric chromosomes in Robertsonian translocations. Ann Hum Genet. 1989;53:49–65.
-
Luciani JM, Guichaoua MR, Mattei A, Morazzani MR. Pachytene analysis of a human with a 13q;14q translocation and infertility. Cytogenet Cel Genet. 1984;38:fourteen–22.
-
Cassuto NG, Le Foll North, Chantot-Bastaraud South, Balet R, Bouret D, Rouen A, et al. Sperm fluorescence in situ hybridization study in nine men carrying a Robertsonian or a reciprocal translocation: relationship between segregation modes and high-magnification sperm morphology examination. Fertil Steri. 2011;96:826–32.
-
ACOG Committee Opinion No. 446: array comparative genomic hybridization in prenatal diagnosis. Obstet Gynecol. 2009;114(5):1161–3.
-
Ferfouri F, Selva J, Boitrelle F, Gomes DM, Torre A, Albert Chiliad, et al. The chromosomal risk in sperm from heterozygous Robertsonian translocation carriers is related to the sperm count and the translocation type. Fertil Steril. 2011;96:1337–43.
-
Mahjoub K, Mehdi M, Brahem Southward, Elghezal H, Ibala South, Saad A. Chromosomal segregation in spermatozoa of five Robertsonian translocation carriers t(13;14). J Assist Reprod Genet. 2011;28:607–13.
-
Rouen A, Pyram K, Pollet-Villard Ten, Hyon C, Dorna M, Marques Southward, et al. Simultaneous cell by cell written report of both DNA fragmentation and chromosomal segregation in spermatozoa from chromosomal rearrangement carriers. J Assist Reprod Genet. 2013. doi:10.1007/s10815-012-9915-7.
-
Machev N, Gosset P, Warter S, Treger M, Schillinger Yard, Viville S. Fluorescence in situ hybridization sperm analysis of 6 translocation carriers provides evidence of an interchromosomal consequence. Fertility and Sterility. 2005;84(2):365–73.
-
Douet-Guilbert N, Bris MJ, Amice 5, Marchetti C, Delobel B, Amice J, et al. Interchromosomal event in sperm of males with translocations: study of 6 cases and review of the literature. Int J Androl. 2005;28:372–9.
-
Britton-Davidian J, Catalan J, da Draca RM, et al. Rapid chromosomal evolution in island mice. Nature. 2000;403:158.
-
Wang W, Lan H. Rapid and parallel chromosomal number reductions in muntjac deer inferred from mitochondrial Dna phylogeny. Mol Biol Evol. 2000;17:1326–33.
-
John B. Chromosome alter and evolutionary change: A critique. In: Atchley WR, Woodruff DS, editors. Evolution and specification: Essays in honor of Thou.J.D. White, Chapter 2. London, New York: Cambridge University; 1981.
-
Benet J, Oliver-Bonet Grand, Cifuentes P, Templado C, Navarro J. Segregation of chromosomes in sperm of reciprocal translocation carriers: a review. Cytogenet Genome Res. 2005;111:281–90.
-
Roux C, Tripogney C, Morel F, Joanne C, Fellmann F, Clavequin MC, et al. Segregation of chromosomes in sperm of Robertsonian translocation carriers. Cytogenet Genome Res. 2005;111:291–6.
-
Rives North, Ravel C, Duchesne V, Siffroi JP, Mousset-Simeon Northward, Mace B. Molecular cytogenetics assay with whole chromosome pigment probes of sperm nuclei from a (xiii;15) Robertsonian translocation carrier. J Hum Genet. 2005;l:360–4.
-
Anahory T, Hamamah S, Andreo B, Hedon B, Claustres Thousand, Sarda P, et al. Sperm segregation analysis of a (13;22) Robertsonian translocation carrier by FISH: a comparison of locus-specific probe and whole chromosome painting. Hum Reprod. 2005;20:1850–4.
-
Lindenbaum RH, Hulten M, McDermott A, Seabright M. The prevalence of translocations in parents of children with regular trisomy 21: a possible interchromosomal effect? J Med Genet. 1985;22:24–viii.
-
Anton E, Blanco J, Vidal F. Meiotic behavior of 3 D;M Robertsonian translocations: segregation and interchromosomal outcome. J Hum Genet. 2010;55:541–54.
-
Ogur G, Van Assche E, Vegetti W, Verheyen Chiliad, Tournaye H, Bonduelle M, et al. Chromosomal segregation in spermatozoa of 14 Robertsonian translocation carriers. Mol Hum Reprod. 2006;12:209–15.
-
Anton E, Vidal F, Blanco J. Role of sperm FISH studies in the genetic reproductive communication of structural reorganization carriers. Hum Reprod. 2007;22:2088–92.
-
Dallapiccola B, Ferranti G, Altissimi D, Colloridi F, Paesano R. Offset-trimester prenatal diagnosis of homozygous (xiv;21) translocation in a fetus with 44 chromosomes. Prenat Diagn. 1989;9:555–eight.
-
Martinez-Castro P, Ramos MC, Rey JA, et al. Homozygosity for a Robertsonian translocation (13q14q) in three offspring of heterozygous parents. Cytogenet Prison cell Genet. 1984;38:310–two.
-
Rajangam South, Michaelis RC, Velagaleti GV, Lincoln S, Hegde S, Lewin S, Tarleton J, Thomas IM , Tharapel AT. Down Syndrome With Biparental Inheritance of der(14q21q) and Maternally Derived Trisomy 21: Confirmation by Fluorescent In Situ Hybridization and Microsatellite Polymorphism Assay. Am J Med Genet Part A. 1997;70:43–7.
-
O'Neill ID. Homozygosity for constitutional chromosomal rearrangements: a systematic review with reference to origin, ascertainment and phenotype. J Hum Genet. 2010;55(9):559–64.
-
Rieseberg LH. Chromosomal rearrangements and speciation. Trends Ecol Evol. 2001;xvi(7):351–viii. doi:10.1016/S0169-5347(01)02187-v.
-
Heng HH. Elimination of contradistinct karyotypes by sexual reproduction preserves species identity. Genome. 2007;l(5):517–24.
-
Horne SD, Chowdhury SK, Heng HH. Stress, genomic adaptation, and the evolutionary trade-off. Forepart Genet. 2014;134(ix):2074–87.
Funding
This work is supported by a scientific inquiry and technology development grant of Guangxi province (#1598012-35)
Availability of data and materials
The dataset(s) supporting the conclusions of this article are available in the binder named raw information for the manuscript at https://www.dropbox.com/sh/izfb6bdmyz43gz7/AAB4nVTovhquFbSqA3kqtTtHa?dl=0
Authors' contributions
B.Westward., J.S. and L.Southward designed the study. B.West. and J.South. analyzed the results and wrote the manuscript. L.S. revised the manuscript. Ten. L. perfomred the sperm FISH. S. X. participated in genetic counseling. N. L. and Z. L. performed array CGH anaysis. Y. Y. and W.W. performed chromosome assay. H. R. perforemed psychiatric interview. All authors read and approved the final manuscript.
Competing interests
The authors declare that they take no competing interests.
Ideals approval and consent to participate
The affected patient were informed nigh the study, and written informed consent, canonical by the Ethical committee of Hubei Maternal and Kid Health Infirmary, was obtained.
Author data
Affiliations
Corresponding authors
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/four.0/), which permits unrestricted utilise, distribution, and reproduction in whatsoever medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Eatables license, and point if changes were made. The Artistic Eatables Public Domain Dedication waiver (http://creativecommons.org/publicdomain/aught/ane.0/) applies to the data fabricated bachelor in this commodity, unless otherwise stated.
Reprints and Permissions
Almost this article
Cite this article
Vocal, J., li, X., Sun, L. et al. A family with Robertsonian translocation: a potential mechanism of speciation in humans. Mol Cytogenet 9, 48 (2016). https://doi.org/10.1186/s13039-016-0255-seven
-
Received:
-
Accepted:
-
Published:
-
DOI : https://doi.org/x.1186/s13039-016-0255-vii
Keywords
- Chromosome karyotype
- FISH
- aCGH
- Robertsonian translocation homozygosity
- Evolution
Source: https://molecularcytogenetics.biomedcentral.com/articles/10.1186/s13039-016-0255-7
0 Response to "What Is the Risk of Having an Abnormal Baby From a Robertsonian Carrier"
Post a Comment